
What Thermal Conductivity Means in Heat Transfer
Thermal conductivity is a fundamental physical property central to thermal physics. It describes how efficiently a material transfers heat energy through thermal conduction. This process involves the movement of energy through a solid without macroscopic motion.
To understand how this applies to everyday objects, looking at the science behind heat conductivity in cookware helps clarify these principles.
Heat conduction occurs when a temperature difference exists. This drives energy from a hotter region to a cooler one to achieve thermal equilibrium. This flow is governed mathematically by Fourier’s law of heat conduction. While thermal transport can occur via convection, conductivity applies specifically to solid materials.
Materials with high thermal conductivities, such as pure metals, move energy rapidly. Conversely, materials with low conductivity act as a thermal insulator. This distinction is vital in engineering and thermophysical property research. It determines how readily temperature will flow through a particular material.
Why Copper Conducts Heat So Efficiently

Copper is a malleable metal and soft metal known for its efficiency. Its ability to act as a conductor of heat stems from its atomic properties. In pure copper, heat is transferred primarily by free electrons.
These electrons facilitate rapid energy transport through the metal lattice. This mechanism allows for quick energy exchanges. It makes copper an efficient conductor of heat and electricity. This efficiency is one of the primary copper cookware benefits valued by manufacturers.
There is a strong correlation between thermal and electrical conductivity. This relationship is described by the Wiedemann-Franz law. Since copper has excellent electrical conductance, it also acts as a superior thermal conductor.
Thermal Conductivity Value of Copper Explained
The thermal conductivity of copper at room temperature is approximately 401 W·m⁻¹·K⁻¹. This is a standardized value used in thermophysical property measurements. Ideally, the thermal conductivity of copper is over 10000 times that of air.
Engineers determine these values using specific tests. Techniques include the steady-state temperature profile method. These tests measure how much power maintains a unit temperature gradient.
Reported values can vary based on purity. Copper samples that are highly refined exhibit better performance than copper alloys. Impurities in alloys often scatter electrons and lower conductivities.
Copper Compared to Other Heat-Conducting Materials
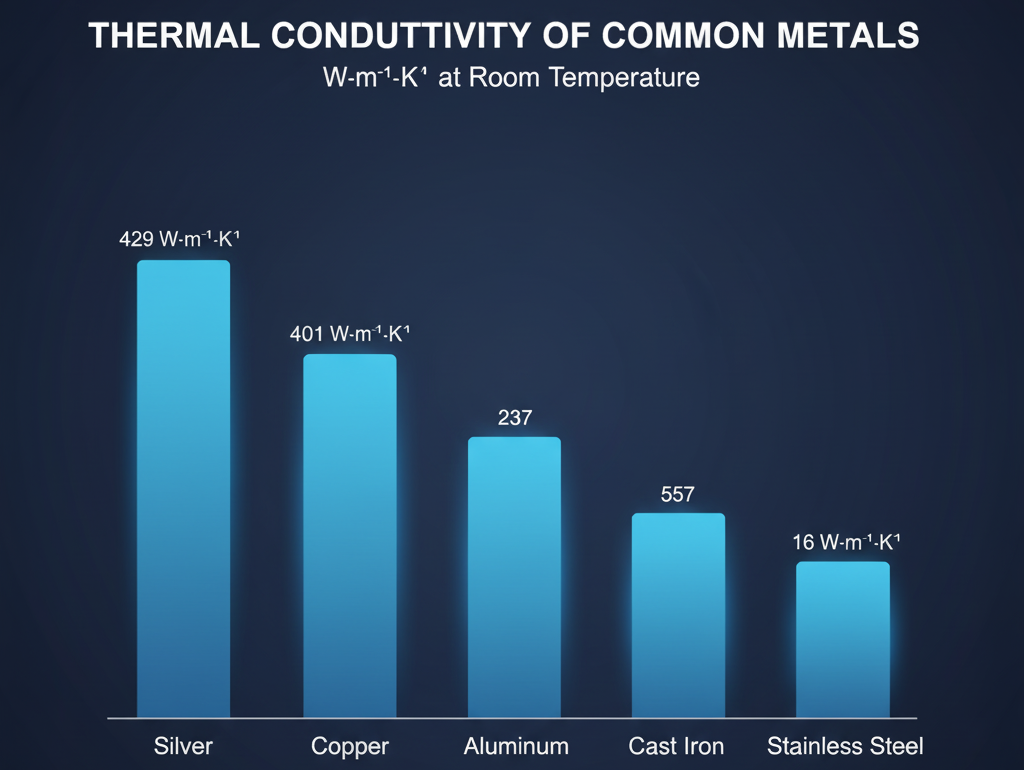
When evaluating different materials, copper stands out near the top. But, it competes with other metallic conductors. Pure silver generally exhibits the highest thermal conductivity, slightly outpacing copper.
Aluminum is a decent thermal conductor but falls short of copper’s metrics. Stainless steel has high thermal resistivity, making it a poor conductor in comparison. When debating is it better to cook with copper or stainless steel, the conductivity gap is the deciding factor.
Cast iron and carbon steel also have lower values than copper. Non-metallic solids generally serve as insulators rather than conductors. In complex designs, engineers often use laminated materials to balance these properties.
Interactive Thermal Conductivity Comparator
Compare heat transfer efficiency across different materials
How Temperature Affects Copper’s Thermal Conductivity

Thermal conductivity changes with temperature. For pure materials like copper, thermal conductivity decreases slightly as high temperatures are reached. This temperature dependence is due to atomic vibrations.
These vibrations, or phonons, interfere with electron paths. At very low temperatures, thermal conductivity increases significantly before dropping off. But, in the standard temperature range of most uses, copper is stable.
This stability is crucial for consistent performance. It ensures that the temperature field remains predictable. This allows for accurate thermophysical simulations in industrial design.
Thermal Diffusivity, Mass, and Resistance
It is important to distinguish between thermal conductivity and thermal diffusivity. Thermal diffusivity combines conductivity, density, and specific heat. It describes how fast a material reacts to a temperature change. This rapid response is why choose copper cookware for temperature-sensitive tasks.
- Thermal Mass: Copper has a relatively high heat capacity for a metal.
- Thermal Resistance: This is the reciprocal of thermal conductance.
- Thermal Effusivity: This defines the ability to exchange thermal energy with surroundings.
Engineers aim to cut thermal resistances in cooling systems. Copper helps bridge thermal gradients effectively. This prevents heat conduction bottlenecks.
Real-World Applications That Rely on Copper

Copper’s properties make it a staple in science and industry. It is essential in heat exchangers and HVAC systems. It is also used in electronics to prevent overheating.
In the culinary world, this precision is critical. It explains why professional chefs like copper pans for sauces and searing. The ability to evenly distribute heat avoids hot spots.
Enthusiasts often seek the best copper frying pans for home kitchens to mimic restaurant results. Additionally, copper foils act as a thermal interface material in tech. They reduce interfacial thermal resistance between computer chips and heat sinks.
Limitations and Trade-Offs
Despite being an excellent conductivity choice, there are limitations. Copper is reactive and can corrode. This affects the surface and potential thermal contact conductance.
In food applications, this reactivity is a concern. Acidic foods can react with the metal. This issue leads consumers to study lined vs unlined copper cookware explained guides before buying.
Cost is another significant factor. Copper is more expensive than different metals like aluminum. It is also a soft metal, which may require alloying for strength.
Common Misconceptions and Advanced Concepts
A common misunderstanding is that conducted heat is the only factor. In reality, thermal contact issues often limit performance. The thermal entrance length in pipes also affects heat transfer rates.
- Geometry: Material thickness matters as much as conductivity.
- Direction: Copper is typically an isotropic material, meaning heat flows equally in all directions.
- Structure: Nanostructured thermoelectrics behave differently than bulk copper.
Evaluating materials function requires looking at the bulk properties. It also involves checking the relaxation length of carriers. Proper analysis ensures the thermal protection system performs as expected.
FAQ Thermal Conductivity of Copper Article
What is the thermal conductivity of copper and what does it mean?
The thermal conductivity of copper is approximately 401 W·m⁻¹·K⁻¹ at room temperature. This means copper can transfer 401 watts of heat energy through one meter of material when there’s a one-degree Kelvin temperature difference. In practical terms, copper conducts heat over 10,000 times better than air, making it one of the most efficient heat-conducting metals available. This property makes copper ideal for applications requiring rapid heat transfer, such as cookware, heat exchangers, and electronics cooling systems.
Why does copper conduct heat better than stainless steel?
Copper conducts heat approximately 25 times better than stainless steel due to fundamental differences in their atomic structure. Copper has abundant free electrons that move rapidly through its metallic lattice, facilitating quick energy transfer. Stainless steel, with a thermal conductivity of only about 16 W·m⁻¹·K⁻¹ compared to copper’s 401 W·m⁻¹·K⁻¹, has high thermal resistivity that impedes heat flow. This significant conductivity gap explains why professional chefs prefer copper cookware for temperature-sensitive cooking tasks where even heat distribution and rapid temperature response are critical.
Does copper or aluminum conduct heat better?
Copper conducts heat significantly better than aluminum. While aluminum is a decent thermal conductor with a value of approximately 237 W·m⁻¹·K⁻¹, copper’s thermal conductivity of 401 W·m⁻¹·K⁻¹ is nearly 70% higher. This means copper transfers heat about 2.3 times more efficiently than aluminum. However, aluminum is often chosen for certain applications because it’s lighter and more cost-effective, even though it doesn’t match copper’s superior thermal performance.
How does temperature affect copper’s ability to conduct heat?
Copper’s thermal conductivity decreases slightly as temperature increases. This happens because higher temperatures cause increased atomic vibrations (called phonons) within the metal, which interfere with the movement of free electrons responsible for heat transfer. However, this decrease is relatively small within the standard temperature range of most applications, and copper remains highly efficient and stable across normal operating conditions. At very low temperatures, copper’s thermal conductivity actually increases significantly before eventually dropping off, but this behavior is mainly relevant for specialized cryogenic applications.
What is the difference between thermal conductivity and thermal diffusivity in copper?
Thermal conductivity measures how well copper transfers heat through its structure, while thermal diffusivity measures how quickly copper responds to temperature changes. Thermal conductivity (401 W·m⁻¹·K⁻¹ for copper) describes the material’s ability to conduct heat energy, whereas thermal diffusivity combines conductivity with the material’s density and specific heat capacity to show the rate of temperature equalization. In practical terms, conductivity tells you how much heat flows through copper, while diffusivity tells you how fast that heat spreads throughout the material. This is why copper cookware heats up quickly and responds rapidly to temperature adjustments during cooking.
Key Takeaways
- Copper’s high thermal conductivity, combined with its strong thermal mass, makes it one of the most effective materials for heat transfer and temperature control. It outperforms most commonly used metals and remains stable across practical temperature ranges.
- Copper is best suited for applications requiring fast, even heat distribution, while alternative materials may be preferable where cost, weight, or chemical resistance are higher priorities.
- Understanding copper within the broader context of materials science, heat transfer physics, and real-world applications allows for better decision-making and more effective thermal design.
External Sources
https://trc.nist.gov/cryogenics/materials/OFHC%20Copper/OFHC_Copper_rev1.htm
https://www.vedantu.com/chemistry/thermal-conductivity-of-copper
https://en.wikipedia.org/wiki/List_of_thermal_conductivities
